Source: Okayama University, Center for Public Affairs and Communications
For immediate release : 16 September 2015
Okayama University research : Simplified boron compound may treat brain tumours
(Okayama, 16 September) Effective and targeted uptake among malignant cells demonstrates the promise of a simplified boron compound for neutron radiation therapy, report researchers at Okayama University and Kinki University.
“Glioblastoma multiforme is the most common malignant central nervous system primary tumour that has been incurable for decades,” explain Hideki Matsui and colleagues at Okayama University Graduate School of Medicine and Kinki University in their recent report. Treatment with ‘boron neutron capture therapy’ (BNCT) has been found to double the duration of patient survival, but finding effective compounds for it has been challenging. Now Michiue and colleagues have demonstrated - both in cell lines and mice - a readily made boron compound that targets and penetrates tumour cells for effective BNCT [1].
The compound BSH is high in boron content and has the advantage of accumulating primarily in tumour tissue rather than healthy tissue. However BSH does not penetrate inside the tumour cells limiting the therapeutic effects of BNCT using it.
Previously Michiue and a collaboration of researchers in Japan demonstrated they could enhance cell penetration by fusing BSH with a large peptide comprising up to 11 amino acid ‘arginine’ groups [2]. However with the large added peptide it was difficult both to produce the compound and track its effects in the body.
Studies of protein transcription aided by a smaller version of the peptide suggested that a compromise in size may allow effective BSH cell penetration without the need for prohibitively complicated synthesis techniques. [3]. The latest results from Michiue and colleagues have now shown that while peptides with just one or two arginine groups were ineffective, three arginine groups were enough to improve cell uptake.
The BSH fused with a peptide of three arginine groups was simpler to produce and they could also track the drug in the body using positron emission tomography. They conclude, “In our present study, we discovered that BSH-3R overcame many obstacles to develop a BSH-peptide suitable for future clinical use.”
Background
Boron neutron capture therapy
One of the challenges with treating glioblastoma multiforme is removing all cancer cells without causing damage to the brain. The destruction of nearby healthy cells is a significant side effect for traditional radiation therapy.
BCNT has attracted particular interest because the radiation caused is highly localised. When boron is delivered to malignant cancer cells, irradiation with low-energy thermal neutrons then triggers boron fission into a high energy alpha particle and lithium atom. While lethal to cells in their direct path the high-energy alpha particle and lithium atom produced cannot travel further than roughly a cell length before their energy is absorbed leaving nearby healthy cells intact.
BNCT has already been used clinically for treating brain tumours, malignant melanoma, head and neck cancers, and hepatoma. Hospital sources for low-thermal-energy neutrons to trigger the fission are in continued development. However clinical use so far has highlighted shortcomings in the boron delivery drugs currently available.
Boron delivery drugs
Previous clinically used drugs for BNCT include an essential amino acid analog p-borono-Lphenylalanine (BPA) and disodium mercaptoundecahydrododecaborate ([B12HnSH]2-2Na+, BSH). BPA readily delivers high boron content to cells but it does not target malignant cells alone and is not taken up by slowly proliferating malignant cells.
In contrast accumulation of BSH is greatly enhanced in tumour regions but it is present between the cells making it less effective for BNCT. Simulations in previous work have highlighted the limitations of BNCT from outside the cell.
Arginine
Arginine is a type of amino acid. Previous work has shown that with an 11-arginine-strong peptide the cancer killing effect of the BSH compound was greater than with a dose 100 times stronger of BSH without the added peptide. However it is difficult to synthesise BSH with the 11-arginine peptide and it cannot be readily tracked to monitor its effects in the body. The results of this latest work resolve some of the practical issues around using arginine groups to improve BSH cell penetration for cancer treatment using BNCT.
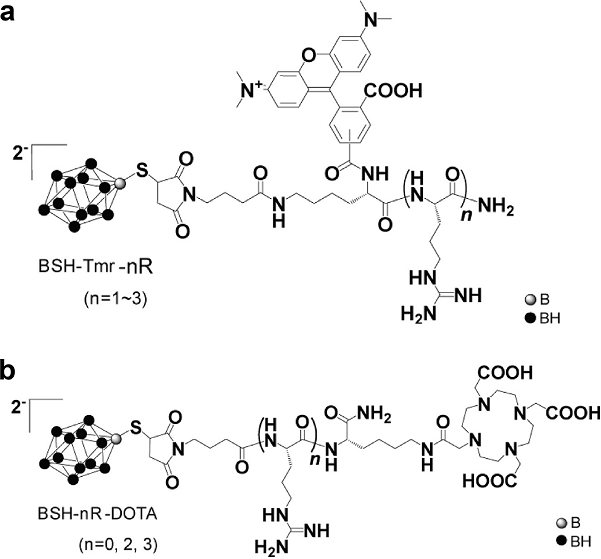
Figure caption
Chemical structures of the readily synthesised BSH fused with comparatively short ‘nR’ peptides, where R is the arginine group and n is the number of arginine groups. When fused with 3R the BSH penetrates the cell for effective BNCT. The chemical is additionally readily fused with a fluorescent moiety, Tmr (a), and DOTA for positron emission tomography scans (b) to track the drug in the body.
References
- Yoshiya Iguchi, Hiroyuki Michiue, Mizuki Kitamatsu, Yuri Hayashi, Fumiaki Takenaka, Tei-ichi Nishiki and Hideki Matsui. Tumor-specific delivery of BSH-3R for boron neutron capture therapy and positron emission tomography imaging in a mouse brain tumor model. (2015) Biomaterials 56 10e17
doi: 10.1016/j.biomaterials.2015.03.061.
http://www.ncbi.nlm.nih.gov/pubmed/25934274 - Hiroyuki Michiue, Yoshinori Sakurai, Natsuko Kondo, Mizuki Kitamatsu, Feng Bin, Kiichiro Nakajima, Yuki Hirota, Shinji Kawabata, Tei-ichi Nishiki, Iori Ohmori, Kazuhito Tomizawa, Shin-ichi Miyatake, Koji Ono and Hideki Matsui. The acceleration of boron neutron capture therapy using multi-linked mercaptoundecahydrododecaborate (BSH) fused cell-penetrating Peptide. (2014) Biomaterials 35 3396e3405
doi: 10.1016/j.biomaterials.2013.12.055.
http://www.ncbi.nlm.nih.gov/pubmed/24452095 - Takashi Hitsuda, Hiroyuki Michiue, Mizuki Kitamatsu, Atsushi Fujimura, Feifei Wang, Takahiro Yamamoto, Xiao-Jian Han, Hiroshi Tazawa, Atsuhito Uneda, Iori Ohmori, Tei-ichi Nishiki, Kazuhito Tomizawa, Hideki Matsui . A protein transduction method using oligo-arginine (3R) for the delivery of transcription factors into cell nuclei. (2012) Biomaterials 33 4665e4672
doi: 10.1016/j.biomaterials.2012.02.049.
http://www.ncbi.nlm.nih.gov/pubmed/22465335
Correspondence to
Assistant Professor Hiroyuki Michiue, M.D., Ph.D.
Department of Physiology, Okayama University
Graduate School of Medicine, Dentistry and
Pharmaceutical Sciences, 2-5-1 Shikata-Cho,
Okayama, 700-8558, Japan.
E-mail: hmichiue@md.okayama-u.ac.jp
Contact information
Public Relations and Information Strategy
E-mail: www-adm(a)adm.okayama-u.ac.jp
For inquiries, please contact us by replacing (a) with the @ mark.
Website: //www.okayama-u.ac.jp/index_e.html
Okayama Univ. e-Bulletin: //www.okayama-u.ac.jp/user/kouhou/ebulletin/
Okayama Univ. e-Bulletin (PDF Issues): //www.okayama-u.ac.jp/user/kouhou/ebulletin/ebulletin.html
About Okayama University (You Tube): https://www.youtube.com/watch?v=iDL1coqPRYI
Okayama University Image Movie (You Tube): https://www.youtube.com/watch?v=_WnbJVk2elA
Okayama University Medical Research Updates (OU-MRU)
Vol.1:Innovative non-invasive ‘liquid biopsy’ method to capture circulating tumor cells from blood samples for genetic testingVol.2:Ensuring a cool recovery from cardiac arrestVol.3:Organ regeneration research leaps forwardVol.4:Cardiac mechanosensitive integratorVol.5:Cell injections get to the heart of congenital defectsVol.6:Fourth key molecule identified in bone developmentVol.7:Anticancer virus solution provides an alternative to surgeryVol.8:Light-responsive dye stimulates sight in genetically blind patientsVol.9:Diabetes drug helps towards immunity against cancerVol.10:Enzyme-inhibitors treat drug-resistant epilepsyVol.11:Compound-protein combination shows promise for arthritis treatmentVol.12:Molecular features of the circadian clock system in fruit fliesVol.13:Peptide directs artificial tissue growth
About Okayama University
Okayama University is one of the largestcomprehensive universities in Japan with roots going back to the Medical Training Place sponsored by the Lord of Okayama and established in 1870. Now with 1,300 faculty and 14,000 students, the University offers courses in specialties ranging from medicine and pharmacy to humanities and physical sciences. Okayama University is located in the heart of Japan approximately 3 hours west of Tokyo by Shinkansen.